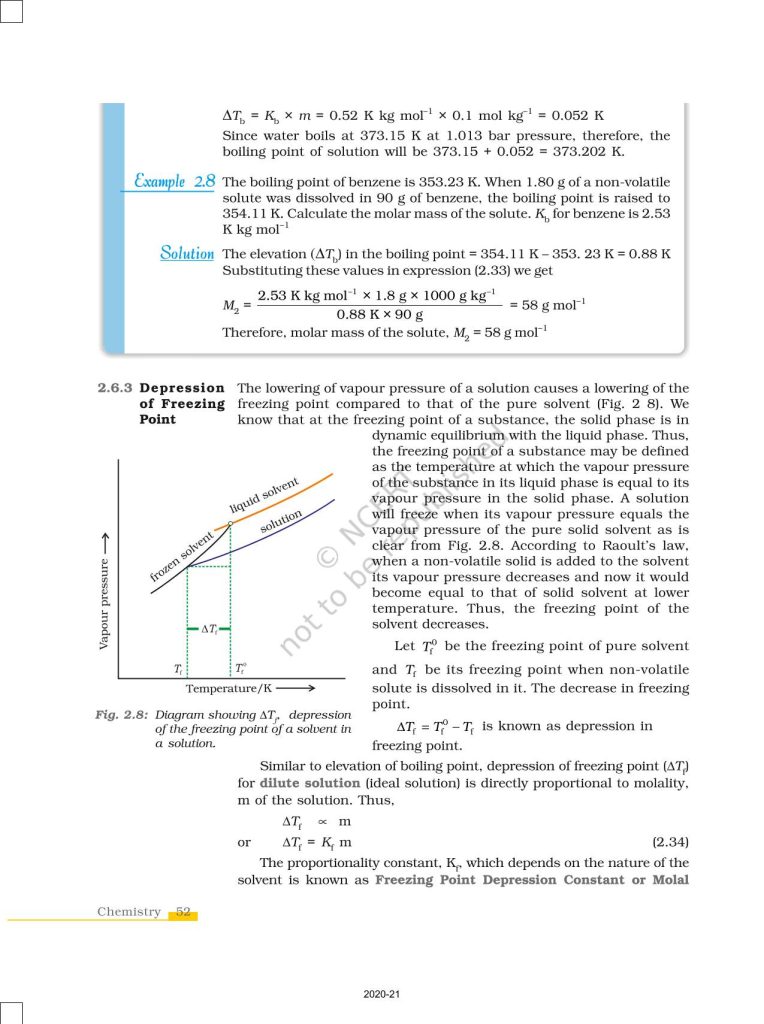
NCERT Book for Class 12 Chemistry Part 1 – (Chapter 2 – Solutions)
Chapter 2 – Solutions
The chapter Solutions explains the properties and behavior of homogeneous mixtures formed when one substance dissolves in another. It introduces different types of solutions based on the physical states of solute and solvent and explains important terms such as solute, solvent, and concentration of solutions. The chapter also discusses various ways to express concentration, including molarity, molality, mass percentage, and mole fraction.
Students also learn about important concepts such as Raoult’s Law, vapor pressure of solutions, ideal and non-ideal solutions, and colligative properties like boiling point elevation, freezing point depression, and osmotic pressure. These topics help explain how the presence of solute affects the physical properties of a solution and are highly important for Class 12 board exams and competitive exams like NEET and JEE.
Solutions Class 12 Chemistry NCERT