5 Simple Steps to Understand the Lewis Structure of PH3 (Phosphine Explained)
A common question in the study or preparation for NEET and test-taking is drawing the Lewis structure of PH3. For those who only want the answer, here’s the direct response:
Table Of Content
The PH3 Lewis structure shows a central phosphorus atom (P) bonded to three hydrogen atoms (H) via three (3) individual covalent bonds. Additionally, the phosphorus atom has an additional pair of unshared electrons.
Therefore, this compound has a trigonal pyramidal molecular shape with phosphorus at the central position similar to ammonia (NH₃).
Many students often struggle with how to draw the Lewis structure for PH3, primarily due to the fact that the steps to arrive at this conclusion can be confusing. However; if you break down the process of constructing a Lewis structure into a few different rules, it then becomes relatively simple to do so.
Let’s go through it together, step-by-step.
🔑 Key Highlights
- The Lewis dot structure illustration of PH3 shows how the electrons are bonded.
- The atomic number of phosphorus is 15, which gives it five electrons to be used for bonding.
- The atomic number of hydrogen is 1, giving it one valence electron.
- In total, there are 8 electrons available to bond PH3.
- Phosphorus can bond to three hydrogen atoms using single covalent bonds.
- Phosphorus has one unshared pair of electrons.
- The geometry of PH3 is trigonal pyramidal.
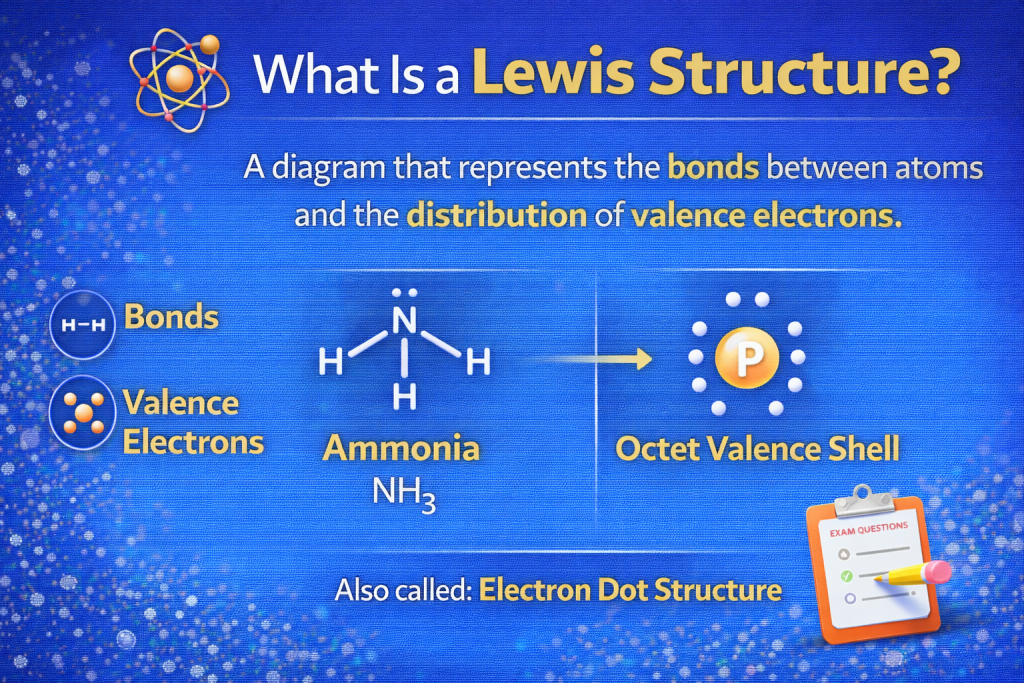
What Is a Lewis Structure?
Before learning how to draw Lewis structure of PH3, it helps to understand what a Lewis structure actually represents.
A Lewis structure (also called an electron dot structure) shows:
- How atoms share electrons
- How covalent bonds form
- Where lone pair electrons exist
In chemistry, these diagrams help you visualize molecular bonding and shape.
American chemist Gilbert N. Lewis introduced this method in 1916, and it remains one of the simplest ways to understand molecular bonding.
External reference:
NECER Question Answers
Understanding the PH₃ Molecule (Phosphine)
The compound PH₃ is called phosphine (IUPAC name: phosphane).
This molecule contains:
- 1 phosphorus atom
- 3 hydrogen atoms
Phosphine is commonly used in:
- Semiconductor manufacturing
- Fumigation pesticides
- Organic chemistry reactions
Because of its toxic nature, phosphine gas must be handled carefully in laboratories.
But in academic chemistry, PH₃ is mainly studied to understand molecular geometry and Lewis structures.
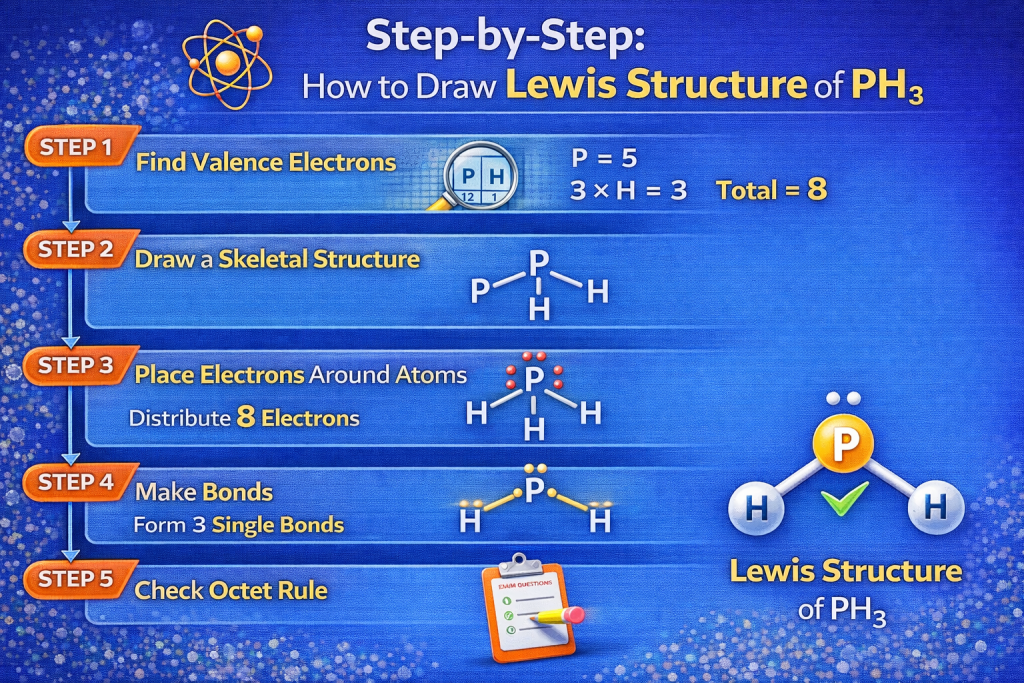
Step-by-Step: How to Draw Lewis Structure of PH3
Let’s break down the process in simple steps.
Step 1: Count the Total Valence Electrons
The first step in drawing the Lewis structure of PH3 is determining the number of valence electrons.
Valence electrons come from the outer shell of atoms.
Phosphorus belongs to Group 15, which means it has 5 valence electrons.
Hydrogen belongs to Group 1, which means each hydrogen atom has 1 valence electron.
Total valence electrons:
- Phosphorus = 5
- Hydrogen (3 × 1) = 3
Total = 8 valence electrons
Step 2: Identify the Central Atom
In Lewis structures, the least electronegative atom usually becomes the central atom.
Hydrogen cannot act as the central atom because it forms only one bond.
Therefore:
👉 Phosphorus becomes the central atom.
The structure layout begins like this:
H
|
H — P — H
Step 3: Form Single Bonds
Each bond represents two electrons.
Phosphorus forms three single covalent bonds with hydrogen atoms.
Each P–H bond uses 2 electrons.
3 bonds × 2 electrons = 6 electrons used
Remaining electrons:
8 – 6 = 2 electrons
Step 4: Place the Remaining Electrons
The remaining 2 electrons become a lone pair on phosphorus.
This lone pair plays a key role in determining the shape of the molecule.
The final Lewis structure appears like this:
..
H — P — H
|
H
The two dots represent the lone pair on phosphorus.
Molecular Geometry of PH₃
Because phosphorus has:
- 3 bonding pairs
- 1 lone pair
The molecular geometry becomes trigonal pyramidal.
This shape forms due to electron pair repulsion, described by the VSEPR theory (Valence Shell Electron Pair Repulsion).
External reference:
Neet Crash Course
Bond angle in PH₃:
≈ 93.5°
This angle is smaller than ammonia (NH₃) because phosphorus atoms are larger.
Why the Lone Pair Matters
Students often ignore the lone pair in the Lewis structure of PH3, but it plays an important role.
Lone pairs cause:
- Repulsion between electrons
- Changes in molecular geometry
- Differences in bond angles
Without the lone pair, PH₃ would have a trigonal planar shape, but because the lone pair exists, it becomes trigonal pyramidal.
Real-World Example: Why Chemists Study PH₃
Understanding how to draw Lewis structure of PH3 is not just for exams.
Chemists use Lewis structures to predict:
- Molecular shape
- Chemical reactivity
- Bond polarity
- Electron distribution
In industrial chemistry, phosphine compounds help manufacture:
- Semiconductors
- Pesticides
- Flame retardants
These applications rely heavily on understanding electron structures of molecules.

Common Mistakes Students Make
Many students preparing for NEET or chemistry exams make these mistakes.
1. Choosing Hydrogen as the Central Atom
Hydrogen can form only one bond, so it must always stay on the outside of the molecule.
2. Forgetting the Lone Pair
Phosphorus has five valence electrons, but only three participate in bonding.
The remaining two electrons form a lone pair.
3. Incorrect Electron Counting
Always remember:
Total electrons in PH₃ = 8 valence electrons
Counting incorrectly leads to the wrong structure.
Study Tip for Chemistry Students
When learning Lewis structures, follow this simple rule:
- Count valence electrons
- Identify the central atom
- Form single bonds
- Complete octets
- Add lone pairs
Practicing this process helps you solve almost any Lewis structure problem quickly.
You can explore more chemistry concepts and NEET preparation guides here:
These resources explain chemistry concepts, molecular structures, and exam questions in a simplified way.
Quick Recap
Let’s summarize the key points.
The Lewis structure of PH3 shows:
- Phosphorus as the central atom
- Three single P–H bonds
- One lone pair on phosphorus
Total valence electrons:
8 electrons
Molecular shape:
✔ Trigonal pyramidal
Understanding how to draw Lewis structure of PH3 helps students learn the fundamentals of chemical bonding and molecular geometry.
Final Thoughts
Chemistry can feel overwhelming when you first encounter molecular structures. Many students look at Lewis diagrams and see only dots and lines.
But once you understand the logic behind electron sharing, the entire system becomes surprisingly clear.
When drawing the Lewis structure of PH3, remember three key ideas:
- Count the electrons
- Place phosphorus in the center
- Don’t forget the lone pair
Once those steps become automatic, Lewis structures stop being confusing and start becoming one of the easiest topics in chemistry.