NCERT Book for Class 12 Chemistry Part 1 – (Chapter 8 – The D And F Block Elements)
Chapter 8 – The D And F Block Elements
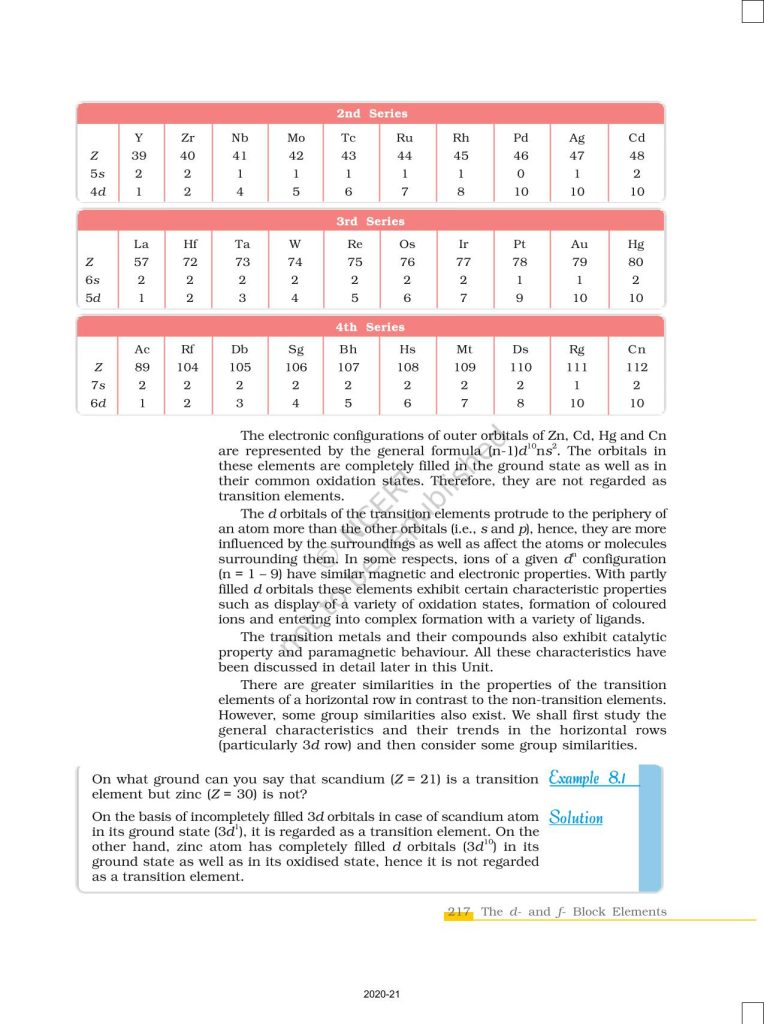
The d- and f-block elements are important groups of metals in the periodic table known for their unique chemical and physical properties. The d-block elements, also called transition elements, are found in the middle of the periodic table (Groups 3–12) and are characterized by the filling of (n−1)d orbitals. They commonly exhibit variable oxidation states, form colored compounds, and act as effective catalysts in many chemical reactions. The f-block elements, known as inner transition elements, include the lanthanides and actinides, where electrons fill the (n−2)f orbitals. Lanthanides are widely used in modern technologies such as magnets, batteries, and lasers, while actinides include radioactive elements like uranium that are important in nuclear energy. Together, these elements play a significant role in industrial applications, advanced materials, and inorganic chemistry. Important for Class 12 board exams and competitive exams like NEET and JEE.
The d and f block Elements NCERT





















- Chapter 9 – Coordination Compounds