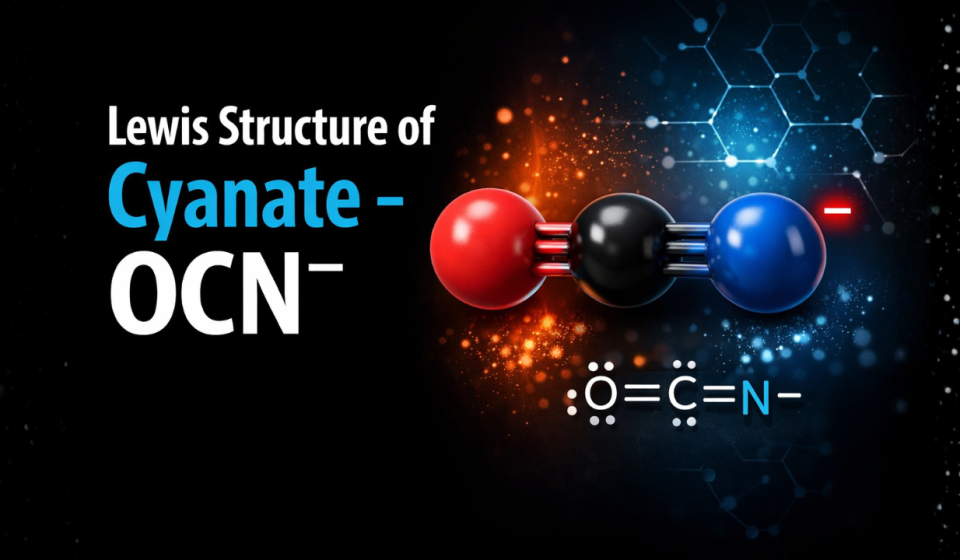
Lewis Structure of Cyanate – OCN‾
🔑 Cyanate Lewis Structure In A Nutshell.
When you are looking to find out the lewis structure of cyanate, here is the answer you are literally seeking:
Table Of Content
- ✨ Key Highlights
- 📘 What is the Cyanate Ion (OCN⁻)?
- 🧠 How to Draw the Lewis Structure of Cyanate Step-by-Step
- 🔗 Step 2: Arrange Atoms
- 🔄 Step 3: Form Initial Bonds
- 🧪 Step 4: Complete Octets
- ⚡ Step 5: Form Multiple Bonds
- 🔁 Resonance in Cyanate Ion
- 📌 Main Resonance Structures
- ⚖️ Formal Charge Calculation
- Formula
- Key Insight
- 📐 Molecular Geometry of Cyanate Ion
- 📈 Where This Concept is Used
- 🔹 1. NEET / JEE Exams
- 🔹 2. Coordination Chemistry
- 🔹 3. Industrial Chemistry
- ⚙️ Best Practices
- 🔗 Internal Learning Links
- 🌍 External References
- ❓ Common Questions
- ❓ How is the lewis structure of cyanate?
- ❓ Number of valence electrons in OCN -?
- ❓ Why is cyanate stable?
- ❓ What is the type of a ligand that is cyanate?
- 🧠 Quick Summary Table
- 🚀 Final Takeaway
- 💬 Conclusion
The resonance structures of the cyanate ion (OCN ‾ ) are:
- N in a Triple bond between Carbon and Nitrogen (C 3 N)
- A single or two bond between Carbon and Oxygen (O=C or O C)
- There were lone pairs handed out to meet the octet rule
- negative charge on Oxygen mostly throughout
✔️ Most stable structure:
👉 O⁻–C≡N
✨ Key Highlights
- ✓ lewis structure of cyanate = OCN‾ with resonance
- ✅ Total valence electrons = 16
- ✅ Carbon is the central atom
- ✔ Structure is in compliance with octet rule
- Oxygen Oxygen is mainly negatively charged
- -1 ✅ Shows multiple valid structures – resonance
📘 What is the Cyanate Ion (OCN⁻)?
You have to be clear on what OCN – is before you can be able to draw the lewis structure of the cyanate ion.
👉 It is a polyatomic ion made of:
- Oxygen (O)
- Carbon (C)
- Nitrogen (N)
👉 It carries a –1 charge
lewis structure of cyanate
This ion is commonly seen in:
- Coordination chemistry
- Chemical industrial processes
- Organic reactions
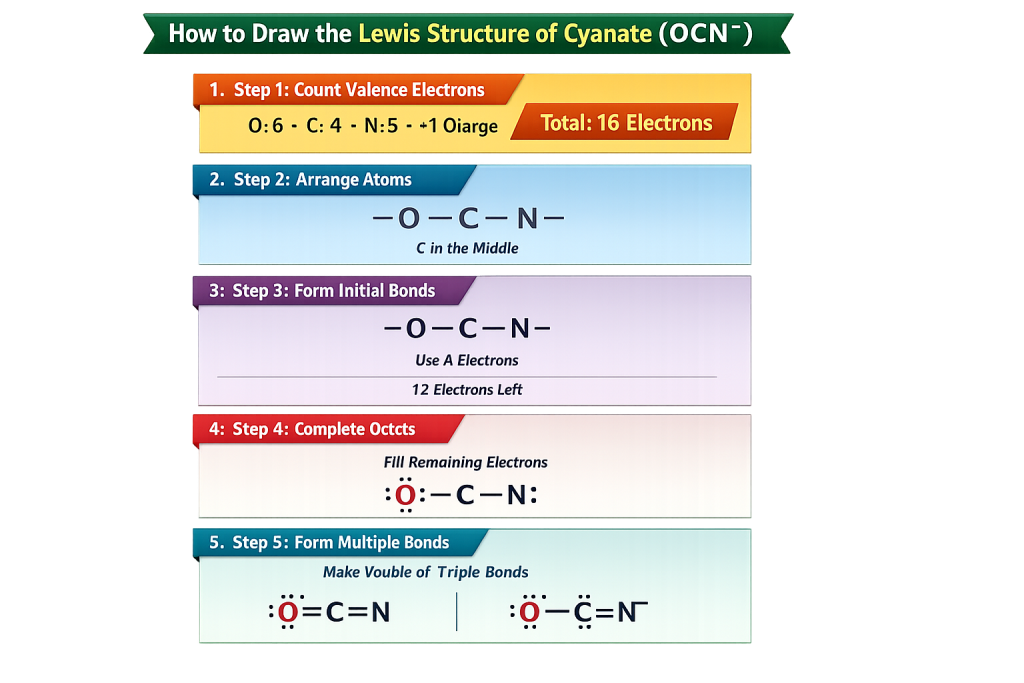
🧠 How to Draw the Lewis Structure of Cyanate Step-by-Step
And we will take this to pieces in a manner that you will remember.
The number of valence electrons can be determined by counting the total number of electrons in the outer electron shell (N).
The total number of valence electrons is equal to the total number of outer electron shell electrons (N).
- Oxygen = 6
- Carbon = 4
- Nitrogen = 5
- The number of additional electrons (negative charge) = 1
👉 Total = 16 valence electrons
🔗 Step 2: Arrange Atoms
The carbon is put in the middle:
O – C – N
Why?
- Carbon easily forms numerous bonds
- It is a link between O and N
🔄 Step 3: Form Initial Bonds
Fuse atoms that are individually bound:
O – C – N
👉 This uses 4 electrons
Remaining = 12 electrons
🧪 Step 4: Complete Octets
Assign out lixons:
- Oxygen gets electrons
- Nitrogen gets electrons
- The octet of carbon is achieved by bonding
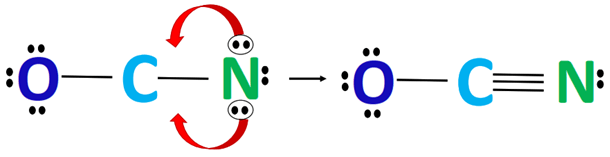
⚡ Step 5: Form Multiple Bonds
To satisfy octet properly:
👉 Form bonds of lone pairs
Final structures include:
- O⁻–C≡N
- O=C=N⁻
🔁 Resonance in Cyanate Ion
This is where the interesting part comes in.
The cyanate ion lewis structure is not in the form of a single structure.
👉 It has resonance forms
📌 Main Resonance Structures:
- O⁻–C≡N (most stable)
- O=C=N⁻
👻 Electrons are delocalized, that is, they are shared by the structure.
⚖️ Formal Charge Calculation
In order to determine the most stable structure:
Formula:
Formal Charge = Valence Electrons – non-bonding + 1/2 Bonding)
Key Insight:
- Oxygen often carries –1 charge
- Carbon remains neutral
- Nitrogen can be charged with structure
👉 Least formally charged structure is the most stable
✔️ So, O⁻–C≡N is preferred
📐 Molecular Geometry of Cyanate Ion
The molecular geometry of cyanate ion is depicted in the figure below.
You might wonder:
What is the shape of this molecule?
- Electron geometry → Linear
- Bond angle → 180°
Why?
👉 There are two regions of electron density formed by carbon.
The thirty-three-year-old woman has an easy time retaining the majority of information she receives (the definition of a memory).
Consider cyanate as a rope that is flexible:
- The electrons do not remain in the same place
- Oxygen and Nitrogen are interchanged
This act of sharing has been termed as resonance.
📈 Where This Concept is Used
🔹 1. NEET / JEE Exams
Direct questions on:
- Lewis structures
- Formal charges
- Resonance
🔹 2. Coordination Chemistry
Cyanate is an ambidentate ligand.
👉 Can bind through:
- Oxygen
- Nitrogen
🔹 3. Industrial Chemistry
Used in:
- Chemical synthesis
- Polymer production
⚙️ Best Practices
The best practices to attract Lewis Structures are described below.
If you want to avoid mistakes:
- ✓ Never leave out valence electrons
- ✔️ Choose correct central atom
- ✔️ Follow octet rule strictly
- ✔️ Check formal charges
- ✔️ Find resonance opportunities
🔗 Internal Learning Links
You can build your chemistry foundations by learning:
- Lewis structures of CO₂, NO₃⁻
- Formal charge calculations
- VSEPR theory
- Chemical bonding basics
🌍 External References
For deeper understanding:
- NEET Crash Course
- Lewis Structures Khan Academy
- Chemical Bonding
- Toppers and educators believe in these.
❓ Common Questions
❓ How is the lewis structure of cyanate?
👉 O⁻–C≡N with resonance forms
❓ Number of valence electrons in OCN -?
👉 16
❓ Why is cyanate stable?
Owing to resonance and charge distribution
because of resonance and charge distribution
❓ What is the type of a ligand that is cyanate?
👉 Ambidentate ligand
🧠 Quick Summary Table
| Property | Value |
|---|---|
| Formula | OCN⁻ |
| Valence Electrons | 16 |
| Shape | Linear |
| Bond Type | Single + Triple |
| Resonance | Yes |
| Charge | –1 |
🚀 Final Takeaway
And you should remember only one thing:
👉 Cyanate has no single structure, it is a resonance hybrid, which is its lewis structure.
✔️ Most stable form: O⁻–C≡N
💬 Conclusion
Initially, it might be confusing to draw Lewis structures such as cyanate. You may have problems with the positioning of electrons, determination of bonds, or in working with charges.
But once you understand:
👉 Count electrons
👉 Follow octet
👉 Check resonance
Everything becomes logical.
This is one of those potent examples which will instruct you in the behavior of the real molecules, not rigid ones in nature, but flexible.
Be a master of this and chemical bonding will not be so difficult.