NCERT Book for Class 12 Physics Part 2 – (Chapter 12 – Atoms)
Chapter 12 – Atoms
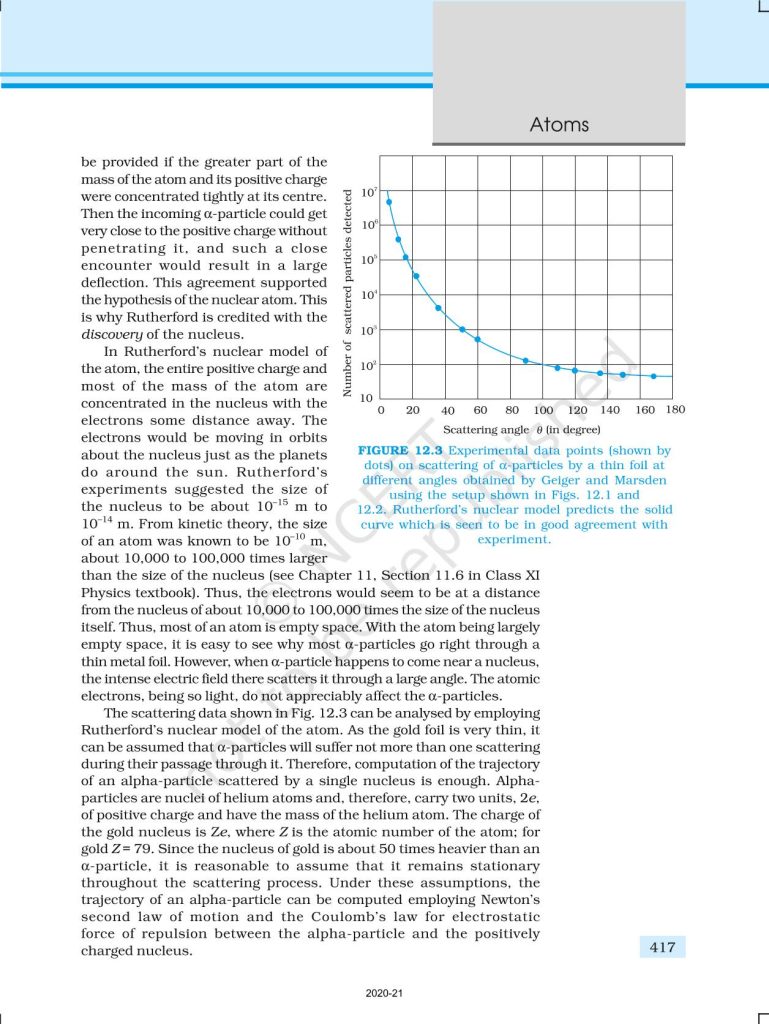
This chapter focuses on the structure and behavior of atoms, introducing key models that explain atomic stability and spectral lines. It begins with early atomic models and highlights the limitations of classical physics, leading to Niels Bohr’s model of the hydrogen atom. Bohr proposed that electrons revolve around the nucleus in fixed energy levels without radiating energy, and transitions between these levels result in the emission or absorption of radiation, producing line spectra. The chapter explains the hydrogen spectrum and its significance in understanding atomic structure. It also introduces the concept of quantization of energy, which plays a crucial role in modern physics. Overall, this chapter lays the foundation for understanding atomic models and the quantum nature of matter. And it is important for Class 12 board exams and competitive exams like NEET and JEE.
Class 12 Atoms NCERT























- Chapter 13 – Nuclei