Charge and Lewis Structure of Nitrate Anion (NO₃⁻)
Charge and Lewis structure of nitrate anion. Here’s the direct answer, no confusion:
👉 The nitrate ion, or NO₃⁻, has a charge of -1.
👉 The nitrate ion has a trigonal planar structure.
👉 The nitrate ion has resonance, or 3 equivalent structures.
Table Of Content
- 🚀 Key Highlights
- 🧪 What is Nitrate Ion (NO₃⁻)?
- 🌍 Real-life importance
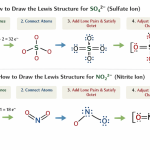
- 🔢 Charge and Lewis Structure of Nitrate Anion (Step-by-Step)
- ✅ Step 1: Count Total Valence Electrons
- ✅ Step 2: Choose the Central Atom
- ✅ Step 3: Draw the Skeleton Structure
- ✅ Step 4: Complete Octet of Oxygen Atoms
- ✅ Step 5: Form Double Bond (Important Step)
- 🔷 Lewis Structure of Nitrate Ion NO₃⁻
- 🧾 Final Structure (Equation Form)
- 💡 What Does This Mean?
- ⚡ Formal Charge of Nitrate Ion
- Formal Charge
- 📐 Geometry of Nitrate Ion
- 🧠 Why Students Get This Wrong
- 💡 Best Practice (Exam Shortcut)
- 🌍 Real-World Use Cases
- 🌱 Agriculture
- 🏭 Industries
- 🧬 Biology
- 🔗 Helpful Resources
- 📘 Internal Links
- 🌐 External Links
- 🤔 Quick Practice Questions
- 🎯 Final Thoughts
Charge and Lewis structure of nitrate anion. Now, you should be able to do this with no problem. However, if you’ve ever felt confused about how to do this, you’re not alone.
We’re going to go over this step by step, as if someone were explaining it to you, not reading from a book.
🚀 Key Highlights
Nitrate ion formula = NO₃⁻
Total valence electrons = 24
Central atom = Nitrogen
Geometry = Trigonal planar
Resonance structures = 3
Overall charge = −1
🧪 What is Nitrate Ion (NO₃⁻)?
Nitrate ion is a polyatomic ion, which means:
👉 It is composed of many atoms joined in a bond, along with a net charge
Nitrate ion is a conjugate base of nitric acid.
🌍 Real-life importance:
Used in fertilizers in the form of ammonium nitrate
Used in soil chemistry/nitrogen cycle
Used in explosives/oxidizing agents
📊 Fact: Nitrate-based fertilizers are responsible for increasing crop yields across the globe.
🔢 Charge and Lewis Structure of Nitrate Anion (Step-by-Step)
Let’s solve it like an exam problem.
✅ Step 1: Count Total Valence Electrons
Formula:
5+(3×6)+1=24 electrons
Nitrogen (N) = 5
Oxygen (O) = 18
Negative charge = +1 electron
👉 Total = 24 electrons
✅ Step 2: Choose the Central Atom
👉 Nitrogen is less electronegative than oxygen
👉 So, Nitrogen is the central atom
✅ Step 3: Draw the Skeleton Structure
Connect nitrogen to 3 oxygen atoms:
O−N−O
(with one more oxygen attached)
3 bonds → 6 electrons used
Remaining = 18 electrons
✅ Step 4: Complete Octet of Oxygen Atoms
Place electrons around oxygen atoms first.
👉 Each oxygen needs 8 electrons
After filling oxygen shells, remaining electrons go to nitrogen.
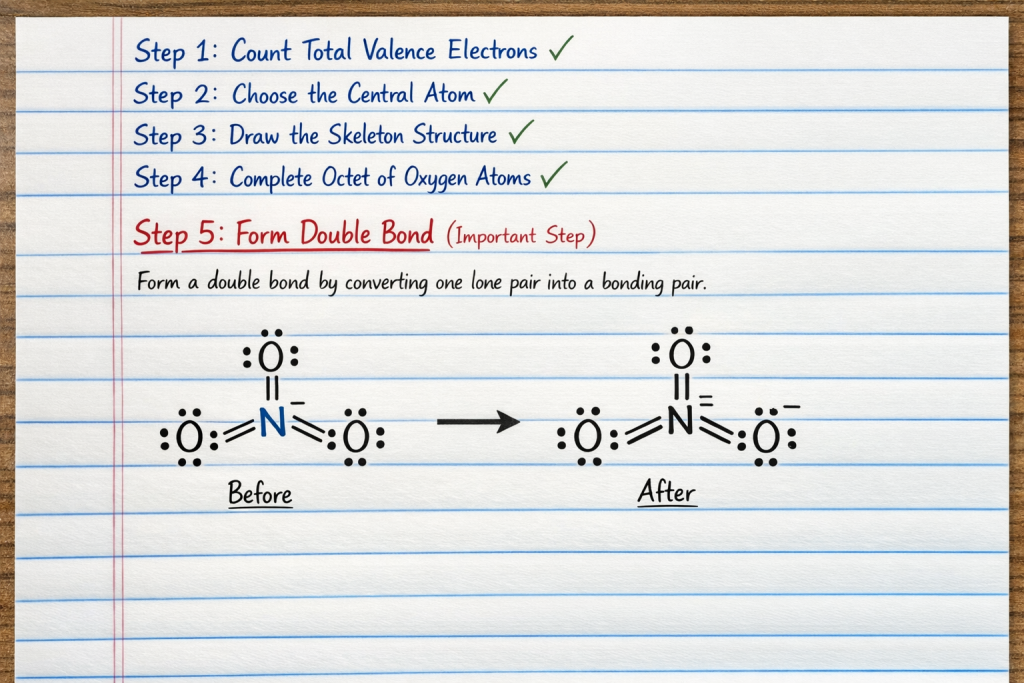
✅ Step 5: Form Double Bond (Important Step)
Here’s where many students get confused.
Nitrogen has only 6 electrons → needs 8
So:
👉 Convert one lone pair from oxygen into a double bond
🔷 Lewis Structure of Nitrate Ion NO₃⁻

🧾 Final Structure (Equation Form)
NO₃‾=O=N−O‾
💡 What Does This Mean?
Charge is delocalized
All N–O bonds are equal
Structure is more stable
⚡ Formal Charge of Nitrate Ion
Let’s check the charge.
Formula:
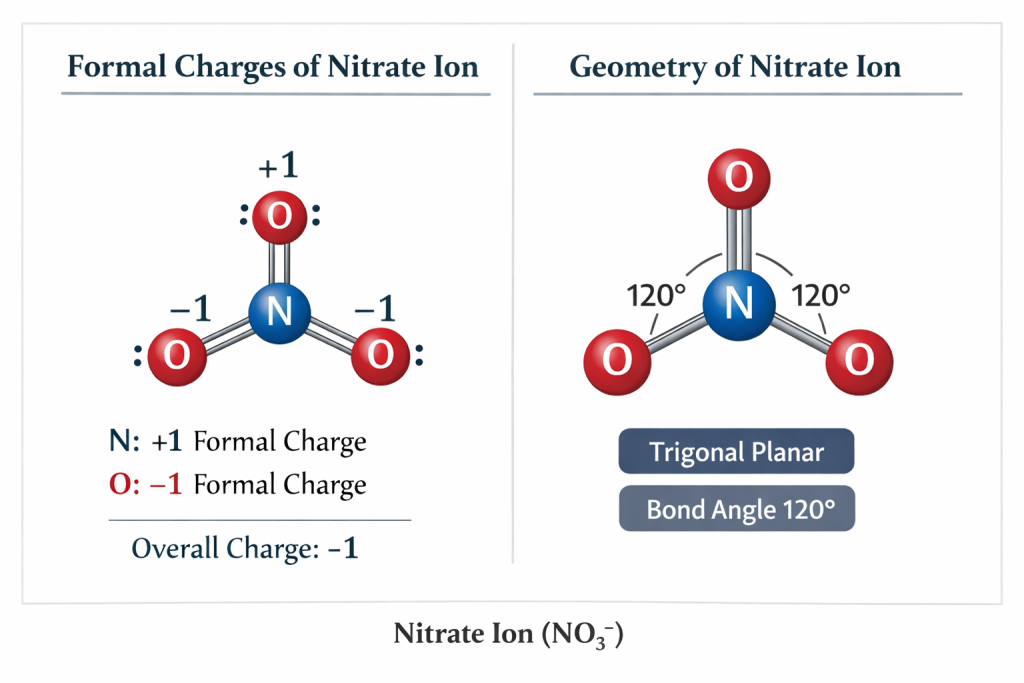
Formal Charge
Formal Charge=Valence−Nonbonding−2/Bonding
👉 Final result:
Nitrogen = 0
Two oxygens = −1
One oxygen = 0
👉 Total = −1
📐 Geometry of Nitrate Ion
Shape = Trigonal Planar
Bond Angle ≈ 120°
👉 Because:
No lone pair on nitrogen
Equal distribution of electrons
🧠 Why Students Get This Wrong
Let’s be honest – the following mistakes are common:
❌ Forgetting to add extra electron from charge
❌ Not forming a double bond
❌ Not considering resonance
❌ Not considering the correct central atom
💡 Best Practice (Exam Shortcut)
Here’s a 5-step shortcut:
Count the electrons
Draw the skeleton
Fill the octet
Form the double bond
Verify the formal charge
👉 This shortcut will work for almost every Lewis structure problem
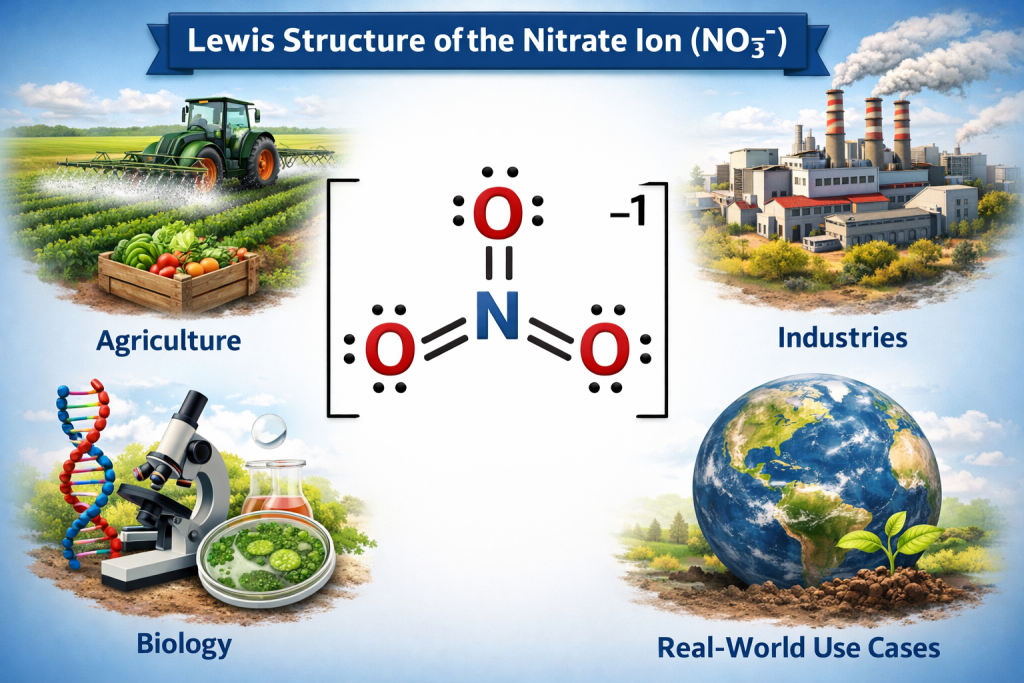
🌍 Real-World Use Cases
Knowing the Lewis structure of the nitrate ion helps us understand its application:
🌱 Agriculture
Nitrate ions are released by fertilizers for plant growth
🏭 Industries
Used for explosives and oxidizers
🧬 Biology
Part of the nitrogen cycle
📊 Quick Summary Table
| Property | Value |
|---|---|
| Formula | NO₃⁻ |
| Total electrons | 24 |
| Charge | −1 |
| Shape | Trigonal planar |
| Resonance | Yes (3 structures) |
🔗 Helpful Resources
📘 Internal Links
Lewis Structure Guide → Learn how bonding occurs
Valence Electrons Guide → Learn how to count them easily
🌐 External Links
🤔 Quick Practice Questions
Why does nitrate ion exhibit resonance?
Why does nitrogen act as the central atom?
What would happen if we do not take into account the charge while doing calculations?
🎯 Final Thoughts
The charge and Lewis Structure of nitrate anion may seem complicated at first.
But if you follow a set procedure…
👉 It makes sense
👉 It becomes logical
👉 It becomes easy
And that is where memorizing ends and understanding begins.