Electron Dot Structure of Propanone
the electron dot structure of propanone is one of those topics that students tend to overthink—but once you get it, it is quite simple really.
the electron dot structure of propanone, also called acetone, is quite simple if you follow a specific method step by step.
Table Of Content
- 🔑 Key Highlights
- 🧠 What is Propanone? (Before Drawing the Structure)
- Propanone (Acetone)
- ✍️ Step-by-Step Guide: Electron Dot Structure of Propanone
- 1️⃣ Identify the Molecular Formula
- 2️⃣ Count the number of valence electrons
- 3️⃣ Arrange the Skeleton Structure
- 4️⃣ Form Bonds (Single + Double)
- 5️⃣ Add Lone Pairs (Critical Step)
- 📊 Final Structure (Visual Understanding)
- 🧠 Why This Structure Works
- ⚠️ Common Mistakes Students Make
- ❌ Mistake 1: Forgetting the Double Bond
- ❌ Single bond between carbon and oxygen
- ❌ Mistake 2: Forgetting the Lone Pairs of Oxygen
- ❌ Mistake 3: Forgetting the Middle Carbon’s Connection to Oxygen
- ❌ Mistake 4: Forgetting the Octet Rule
- 🧠 Best Practices to Draw Lewis Structure of Propanone
- ✔️ 1. Always Count Electrons First
- ✔️ 2. Draw Skeleton Before Bonds
- ✔️ 3. Check Octet Rule
- ✔️ 4. Practice Similar Molecules
- 📊 Real-Life Applications (Why This Matters)
- 🧪 1. Chemistry & Lab Work
- 💻 2. Drug Design & Research
- 🏭 3. Industrial Use
- 🔗 NEET Crash Course
- 🔗 Internal Learning Suggestions
- 📈 Why This Topic Helps Your Career
- 🤔 Quick Self-Test
- 🧾 Summary
- 🎯 Final Thought
👉 Quick answer:
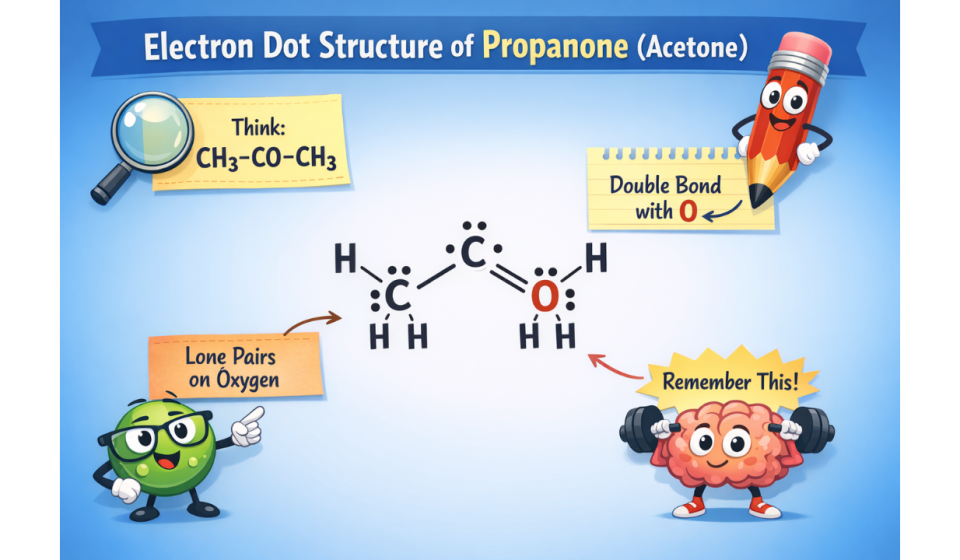
the structure is CH₃–CO–CH₃, and it has a double bond between the carbon and oxygen and two lone pairs of electrons on the oxygen atom.
Okay, now let’s look at it in a way that you will actually remember it for your exams.
NCERT Chemistry Question & Answer
🔑 Key Highlights
- Electron Dot Structure of Propanone: CH₃–CO–CH₃
- Central carbon forms a double bond: C=O
- Oxygen has 2 lone pairs
- Total Formula: C₃H₆O
- Follows the octet rule (except for hydrogen)
- Also known as acetone
- Simplest ketone
🧠 What is Propanone? (Before Drawing the Structure)
Before we get into the electron dot structure for propanone, let’s first try to get a general idea of what we’re working with.
Propanone (Acetone):
- Chemical Formula: C₃H₆O
- Family: Ketones
- Commonly used as: Nail Polish Remover
- Used in: Laboratories
👉 Real-life connection: Every time you use a nail polish remover, you’re basically using propanone.
✍️ Step-by-Step Guide: Electron Dot Structure of Propanone
Okay, now let’s get cracking! We’re not taking any shortcuts here! This is exactly how you would solve it for an exam too!
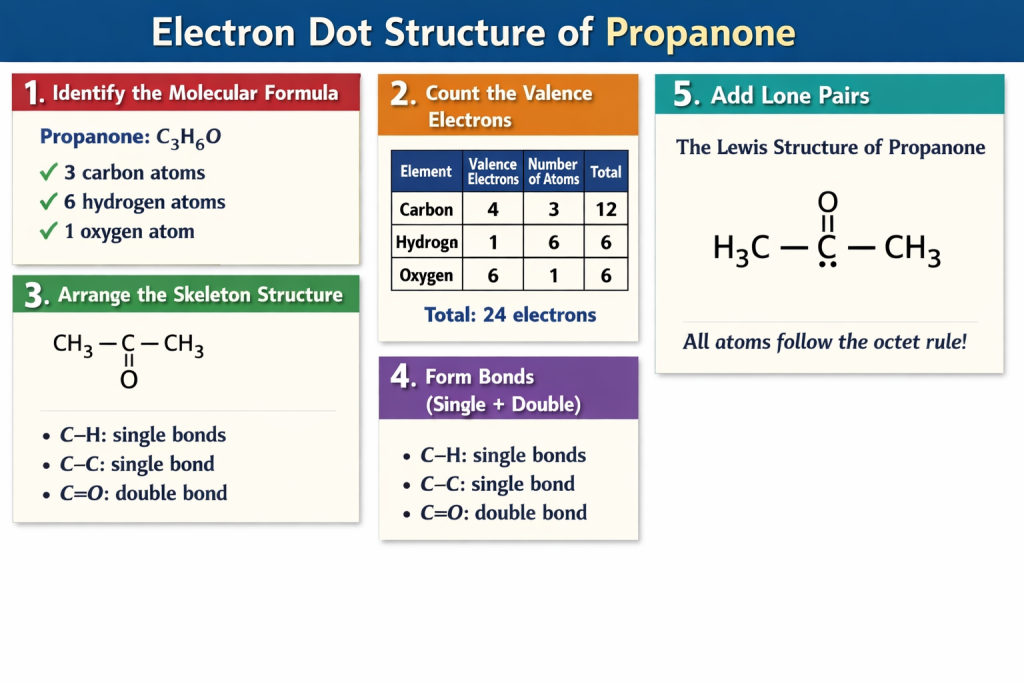
1️⃣ Identify the Molecular Formula
Propanone: C₃H₆O
This tells you:
- 3 carbon atoms
- 6 hydrogen atoms
- 1 oxygen atom
2️⃣ Count the number of valence electrons
This is the most important step for the electron dot structure for propanone!
| Element | Valence Electrons | Number of Atoms | Total |
|---|---|---|---|
| Carbon | 4 | 3 | 12 |
| Hydrogen | 1 | 6 | 6 |
| Oxygen | 6 | 1 | 6 |
| Total | 24 electrons |
👉 You are working with 24 valence electrons
3️⃣ Arrange the Skeleton Structure
The carbon atom will always be in the center.
👉 Arrange the structure like so:
CH₃ – C – CH₃
Then, we’ll put the oxygen atom next to the carbon atom in the center:
CH₃ – C – CH₃
||
O
4️⃣ Form Bonds (Single + Double)
- C-H: single bond
- C-C: single bond
- C=O: double bond
👉 This double bond gives propanone its status as a ketone
5️⃣ Add Lone Pairs (Critical Step)
The Lewis structure of propanone can now be drawn:
Oxygen has 2 lone pairs: •• ••
All atoms are following the octet rule
📊 Final Structure (Visual Understanding)
Here’s a simple structure of propanone:
🧠 Why This Structure Works
If you’re wondering “Why double bond?” here’s the answer:
- Carbon has 8 electrons to share
- Oxygen has 8 electrons to share
- A double bond satisfies both atoms
- 👉 And that’s why the electron dot structure of propanone is stable!
⚠️ Common Mistakes Students Make
Let’s correct our mistakes before we lose our marks!
❌ Mistake 1: Forgetting the Double Bond
What’s gone wrong?
❌ Single bond between carbon and oxygen
What’s the correction?
👉 Double bond between carbon and oxygen
❌ Mistake 2: Forgetting the Lone Pairs of Oxygen
Oxygen needs 2 lone pairs of electrons
❌ Mistake 3: Forgetting the Middle Carbon’s Connection to Oxygen
The middle carbon atom needs to connect to oxygen
❌ Mistake 4: Forgetting the Octet Rule
Remember: Every atom (except hydrogen) needs 8 electrons\
🧠 Best Practices to Draw Lewis Structure of Propanone
To get good marks in this topic, you need to:
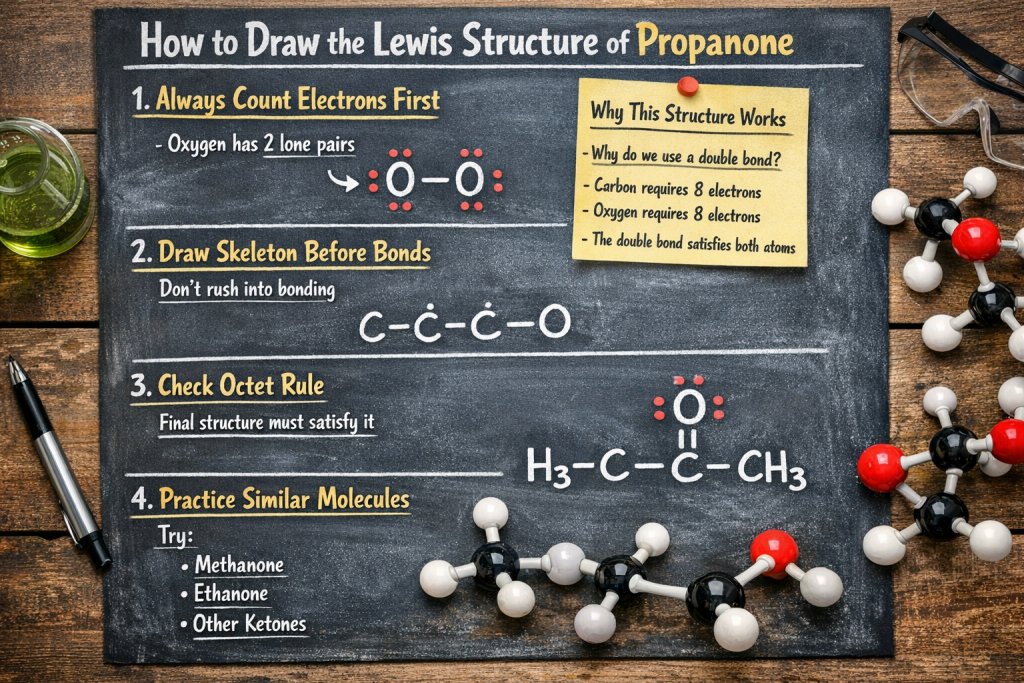
✔️ 1. Always Count Electrons First
👉 Oxygen has 2 lone pairs
🧠 Why This Structure Works
You might be wondering: “Why do we use a double bond?” Well, here’s the answer:
- Why do we use a double bond?
- Carbon requires 8 electrons
- Oxygen requires 8 electrons
- The double bond satisfies both atoms
- 👉 This is why the electron dot structure of propanone is stable
✔️ 2. Draw Skeleton Before Bonds
Don’t rush into bonding
✔️ 3. Check Octet Rule
Final structure must satisfy it
✔️ 4. Practice Similar Molecules
Try:
- Methanone
- Ethanone
- Other Ketones

📊 Real-Life Applications (Why This Matters)
You might think this is just theory—but it’s not.
🧪 1. Chemistry & Lab Work
Understanding the electron dot structure of propanone is useful in:
- Predicting reactions
- Understanding bonding
- Chemical synthesis
💻 2. Drug Design & Research
Understanding the structure of molecules is vital in:
- Pharmaceuticals
- Biochemistry
- Material science
🏭 3. Industrial Use
Propanone is used in:
- Cleaning agents
- Paint thinners
- Plastic production
🔗 NEET Crash Course
To learn more:
These are trusted sources for chemistry
🔗 Internal Learning Suggestions
You can learn:
- Lewis structures of other organic compounds
- Chemical bonding basics
- Valence electrons basics
(Useful to link your content or website)

📈 Why This Topic Helps Your Career
This topic develops:
- Analytical skills
- Problem-solving skills
- Scientific skills
In industries like:
- Chemical engineering
- Pharmacy
- Research
- Data modeling
👉 Proper basics lead to proper careers.
🤔 Quick Self-Test
You can do:
👉 How many lone pairs are there in oxygen in propanone?
✔ Answer: 2
👉 What kind of bond exists between C and O?
✔ Answer: Double bond
🧾 Summary
- electron dot structure of propanone = CH₃–CO–CH₃
- Carbon in the center has a double bond with oxygen
- Oxygen has 2 lone pairs
- Follows the octet rule too
- Also known as acetone
🎯 Final Thought
At first glance, it might look a bit confusing to draw structures like these. You might take your time or think twice before starting to draw. This is perfectly normal.
But once you go through all these steps to draw a Lewis structure, it all becomes logical to you.
The next time someone asks you to draw the Lewis structure of propanone, you won’t think twice. You’ll do it correctly.