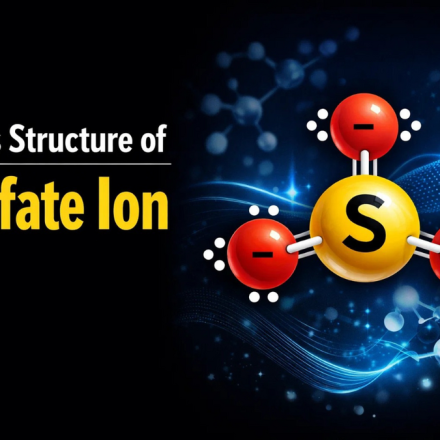
How to Draw Lewis Structure for SO₄²‾ and NO₂‾
How to draw Lewis structure for SO₄²‾ and NO₂‾ this is one of those things that looks super scary until you realize the pattern behind it.
Table Of Content
- 🚀 Key Highlights
- ✅ Step 1: Count Total Valence Electrons (Equation Method)
- 🔹 For SO₄²‾ (Sulfate Ion)
- 🔹 For NO₂‾ (Nitrite Ion)
- 💡 Pro Tip
- ✅ Step 2: Choose the Central Atom
- ✅ Step 3: Draw the Basic Skeleton
- ✅ Step 4: Distribute Remaining Electrons
- 🔥 Real Insight (What Toppers Do Differently)
- 🧪 Lewis Structure of Sulfate Ion and Nitrite Ion (Final Forms)
- 🔷 Lewis Structure of Sulfate Ion (SO₄²‾)
- 🧾 Final Equation Form
- ✔ Key Observations
- 🌍 Real-World Connection
- 🔷 Lewis Structure of Nitrite Ion (NO₂‾)
- 🧾 Final Equation Form
- ✔ Key Observations
- 🌍 Real-World Connection
- ⚠️ Common Mistakes You Must Avoid
- 💡 Best Practice (Works Every Time)
- 🔗 Helpful Resources
- 📘 Internal Guides
- 🌐 External Resources
- 🎯 Final Thoughts
Ever stared at a chemistry question and thought, “What in the world do I do first?” – well, you’re not alone, friend! This is one of those tricky parts for people to understand because they’re trying to memorize things instead of understanding them.
Let’s fix that, shall we? With this guide, you’ll be able to solve any Lewis structure question, particularly the Lewis structure for sulfate ion and nitrite ion, with ease!
🚀 Key Highlights
- Learn how to draw the Lewis Structure of SO₄²‾ and NO₂‾ in a step-by-step manner.
- Learn the calculation of valence electrons in a simplified manner using equations.
- Learn how to avoid the #1 common mistake students do (when drawing the Lewis Structure).
- Learn how to master the drawing of resonance structures.
🧪 How to Draw Lewis Structure for SO₄²‾ and NO₂‾ (Step-by-Step)
Let’s break this into a method you can reuse in exams.

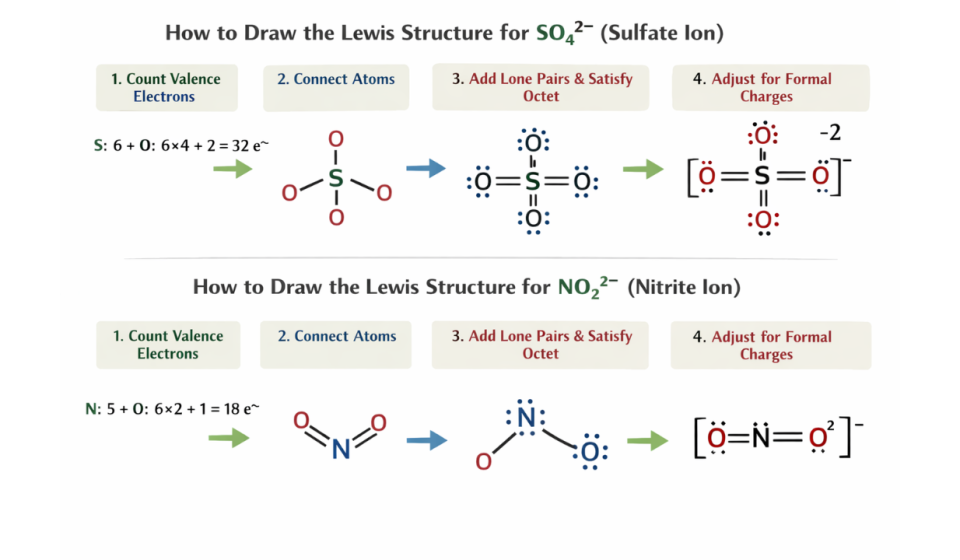
✅ Step 1: Count Total Valence Electrons (Equation Method)
This is where most students either win or lose their marks.
🔹 For SO₄²‾ (Sulfate Ion)
6 + (4 x 6) + 2 = 32 electrons
Sulfur = 6
Oxygen = 24
Charge = +2 electrons
👉 Total = 32 electrons
🔹 For NO₂‾ (Nitrite Ion)
5 + (2 x 6) + 1 = 18 electrons
Nitrogen = 5
Oxygen = 12
Charge = +1 electron
👉 Total= 18 electrons
💡 Pro Tip:
Negative Charge = Add electrons
Positive Charge = Subtract electrons
✅ Step 2: Choose the Central Atom
Always select:
👉 Least electronegative atom except Hydrogen
SO₄²‾ → Sulfur is central atom
NO₂‾ → Nitrogen is central atom
✅ Step 3: Draw the Basic Skeleton
Connect all atoms using single bonds.
Example:
SO₄²‾ → Sulfur is connected to 4 Oxygen atoms
NO₂‾ → Nitrogen is connected to 2 Oxygen atoms
Each bond is composed of 2 electrons
✅ Step 4: Distribute Remaining Electrons
After making bonds:
First, fill in the outer atoms. In this case, we have oxygen.
Then, fill in the central atom.
👉 Remember to always follow the octet rule, which is 8 electrons.
🔥 Real Insight (What Toppers Do Differently)
- Top performers do not randomly distribute electrons. They:
- Give priority to oxygen atoms.
- Carefully keep track of the number of remaining electrons.
- Make sure to remember the octet rule.
- This is why they never get
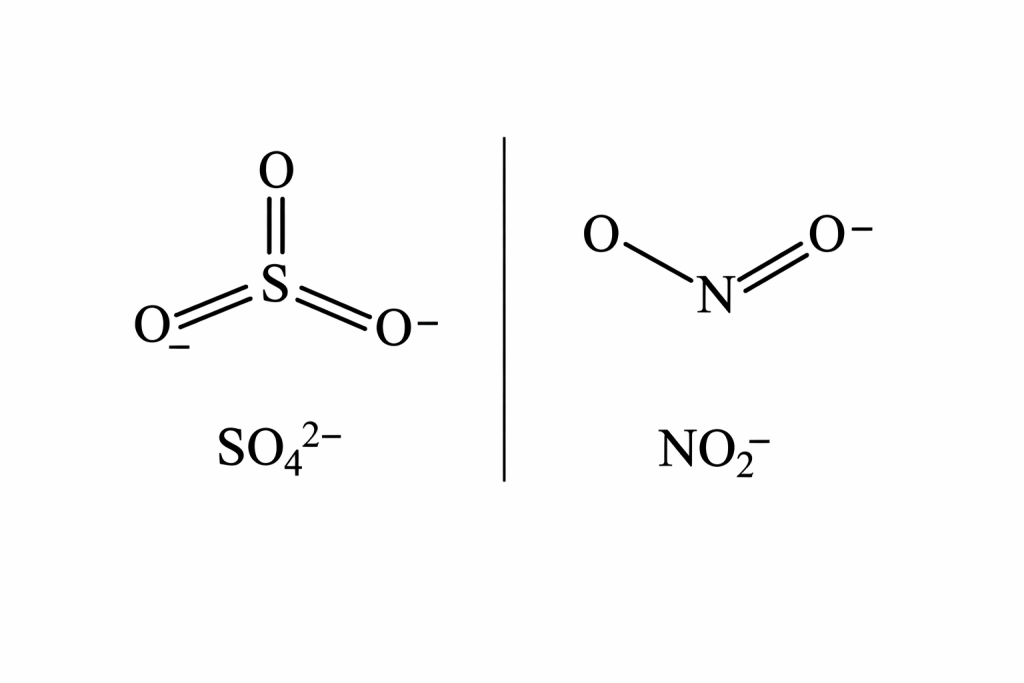
🧪 Lewis Structure of Sulfate Ion and Nitrite Ion (Final Forms)
🔷 Lewis Structure of Sulfate Ion (SO₄²‾)
🧾 Final Equation Form
SO₄²‾=O=S(=O)(O‾)(O‾)
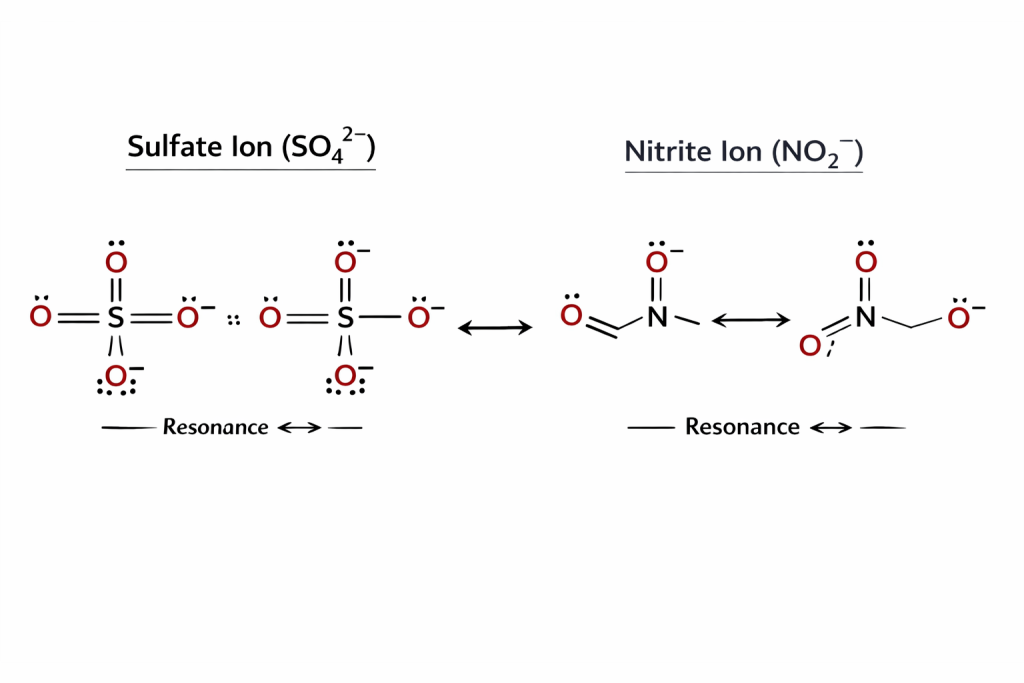
✔ Key Observations
- 2 double bonds (S=O)
- 2 single bonds (S–O⁻)
- Sulfur expands its octet
- Shows resonance
🌍 Real-World Connection
- Sulfate ions are all around us:
- In fertilizers (ammonium sulfate)
- In water treatment
- In biological systems
- Understanding the structure of the sulfate ion has many practical applications.
🔷 Lewis Structure of Nitrite Ion (NO₂‾)
🧾 Final Equation Form
NO₂‾=O=N−O‾
✔ Key Observations
- 1 double bond
- 1 single bond
- 1 lone pair on nitrogen
- Charge is delocalized
🌍 Real-World Connection
Nitrite ions have the following applications:
- In food preservation
- In medical chemistry, it causes vasodilation
- In the nitrogen cycle in soil
⚠️ Common Mistakes You Must Avoid
Face it, these are the reasons you’re losing marks:
❌ Don’t forget to add electrons for the negative charge!
❌ Don’t break the octet rule!
❌ Don’t forget double bonds!
❌ Don’t forget resonance!
💡 Best Practice (Works Every Time)
This works every time, 90% guaranteed! Here’s how you do it:
✔ Count electrons
✔ Draw skeleton
✔ Fill octet
✔ Use double bonds
✔ Check formal charge
📊 Quick Comparison Table
| Feature | SO₄²‾ | NO₂‾ |
|---|---|---|
| Total electrons | 32 | 18 |
| Central atom | Sulfur | Nitrogen |
| Double bonds | 2 | 1 |
| Resonance | Yes | Yes |
🔗 Helpful Resources
📘 Internal Guides
Learn bonding basics → Chemical Bonding Complete Guide
Practice questions → NEET Lewis Structure Mock Test
🌐 External Resources
🎯 Final Thoughts
- How to draw Lewis structure for SO₄²‾ and NO₂‾ becomes easy when you stop memorizing and start understanding.
- It looks like a lot of work at first.
- Too many steps.
- Too many rules.
- Too much confusion.
- But when you try this out a few times…
- 👉 You’ll be solving structures faster than you thought possible
- 👉 You’ll know your mistakes in an instant
- 👉 You’ll even begin to enjoy your chemistry problems
That’s the big secret most people miss.