Draw the Lewis Structure for Carbonate Ion (CO₃²⁻)
The carbonate ion (CO₃²⁻) is an important polyatomic ion commonly studied in chemistry. It appears in many compounds such as calcium carbonate (CaCO₃) found in limestone, chalk, and marble. A common question in chemistry exams is: “Draw the Lewis structure for carbonate ion (CO₃²⁻).”
Table Of Content
- What Is a Lewis Structure?
- Understanding the Carbonate Ion (CO₃²⁻)
- Key Properties of Carbonate Ion
- Step-by-Step Explanation to Draw the Lewis Structure
- Step 1: Count the Valence Electrons
- Step 2: Identify the Central Atom
- Step 3: Draw Single Bonds
- Step 4: Distribute Remaining Electrons
- Step 5: Form a Double Bond
- Resonance in Carbonate Ion
- What Is Resonance?
- Final Answer
- Additional Notes
- Important Points About Lewis Structures
- Real-Life Importance of Carbonate Ion
To draw a Lewis structure, we represent atoms, bonding electrons, and lone pairs using dots and lines. This helps us understand how atoms share electrons and how molecules or ions are structured.
The carbonate ion carries a −2 charge and consists of one carbon atom bonded to three oxygen atoms. The arrangement of atoms forms a trigonal planar structure, which means the atoms lie in the same plane around the carbon atom.
Students preparing for chemistry exams can find more explanations and solved questions at chennaineet.
What Is a Lewis Structure?
A Lewis structure (also called a Lewis dot diagram) is a diagram that shows:
- The bonding between atoms
- The valence electrons involved in bonding
- The lone pairs of electrons
Electrons are usually represented as dots, and chemical bonds are represented as lines between atoms.
Lewis structures are used to understand:
- Molecular bonding
- Electron distribution
- Molecular geometry
Understanding the Carbonate Ion (CO₃²⁻)
The carbonate ion is a polyatomic ion, meaning it contains more than one atom bonded together and carries a net charge.
Key Properties of Carbonate Ion
- Chemical formula: CO₃²⁻
- Molecular mass: 60 u
- Geometry: Trigonal planar
- Charge: −2
- Central atom: Carbon
The carbonate ion is also the conjugate base of bicarbonate ion (HCO₃⁻), which itself is the conjugate base of carbonic acid (H₂CO₃).
Step-by-Step Explanation to Draw the Lewis Structure
Step 1: Count the Valence Electrons
First, calculate the total number of valence electrons.
- Carbon (C) → 4 electrons
- Oxygen (O) → 6 electrons × 3 = 18 electrons
- Additional charge (2⁻) → 2 electrons
Total valence electrons:
4 + 18 + 2 = 24 electrons
Step 2: Identify the Central Atom
In the carbonate ion:
- Carbon acts as the central atom
- Three oxygen atoms surround the carbon atom.
Step 3: Draw Single Bonds
Connect carbon to each oxygen atom with single bonds.
This uses 6 electrons (3 bonds × 2 electrons).
Step 4: Distribute Remaining Electrons
The remaining electrons are placed as lone pairs on the oxygen atoms to complete their octet.
Step 5: Form a Double Bond
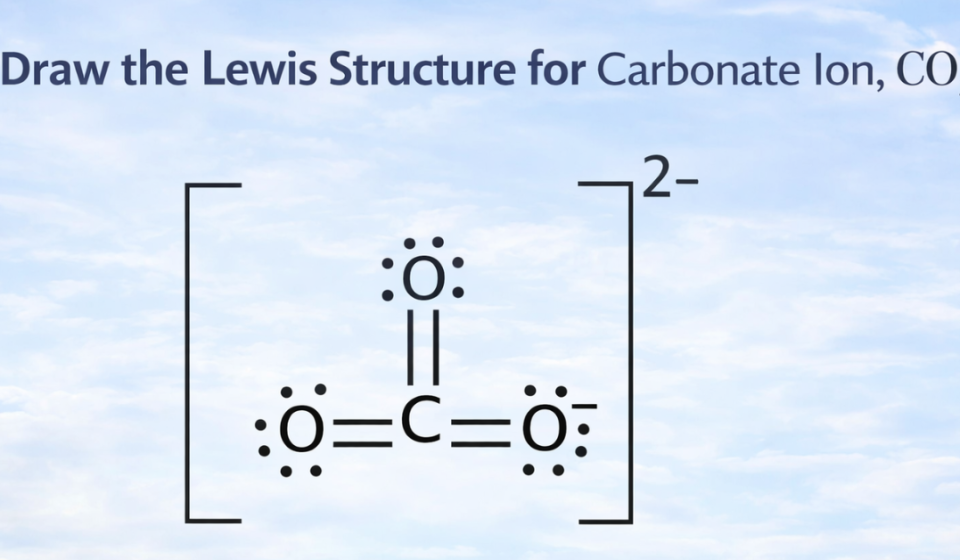
To satisfy the octet rule for carbon, one oxygen atom forms a double bond with carbon.
So the structure includes:
- One double bond (C=O)
- Two single bonds (C–O⁻)
The two oxygen atoms with single bonds carry negative charges.
Resonance in Carbonate Ion
At first glance, the carbonate ion appears to have:
- Two single bonds
- One double bond
However, experiments show that all three carbon–oxygen bonds have equal lengths.
This happens due to resonance.
What Is Resonance?
Resonance means that electrons are delocalized, and the double bond moves between the oxygen atoms.
Thus, there are three equivalent resonance structures, where the double bond shifts among the three oxygen atoms.
As a result:
- All C–O bonds become identical
- Each bond has partial double-bond character
Final Answer
The Lewis structure of carbonate ion (CO₃²⁻) contains:
- One carbon atom in the center
- Three oxygen atoms surrounding it
- One double bond between carbon and oxygen
- Two single bonds between carbon and oxygen atoms carrying negative charges
- A total charge of −2
Due to resonance, the double bond shifts between oxygen atoms, making all three bonds equal in length.
Additional Notes
Important Points About Lewis Structures
- Lewis structures are also called:
- Lewis dot diagrams
- Electron dot structures
- Lewis electron structures
- They help visualize:
- Bonding electrons
- Lone pairs
- Molecular geometry
Real-Life Importance of Carbonate Ion
The carbonate ion is found in many important substances such as:
- Limestone (CaCO₃)
- Baking soda reactions
- Marine shells and coral
- Carbonate minerals
Understanding its Lewis structure helps explain bonding, resonance, and molecular stability in chemistry.
For more chemistry explanations and solved questions, visit chennaineet.