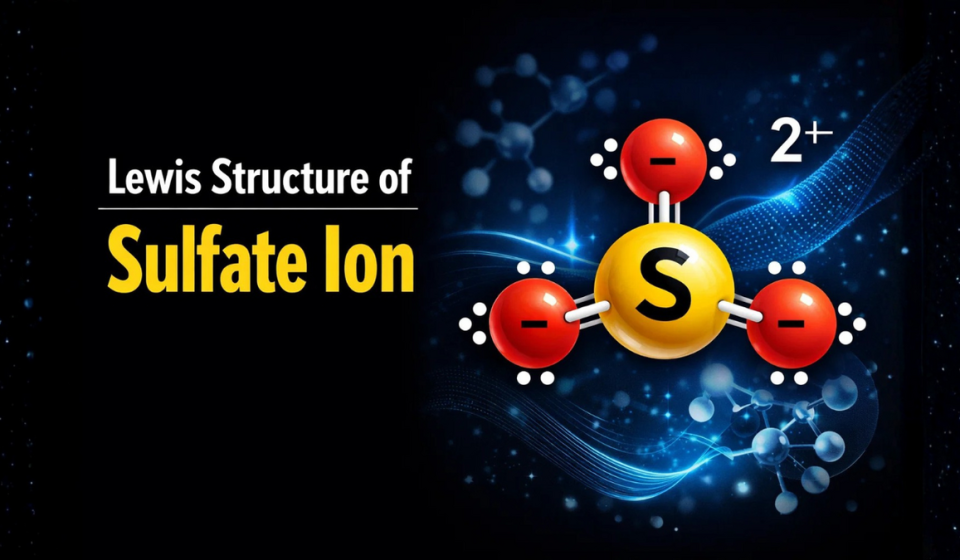
Lewis Structure of Sulfate Ion
Quick Answer (Get It Instantly!)
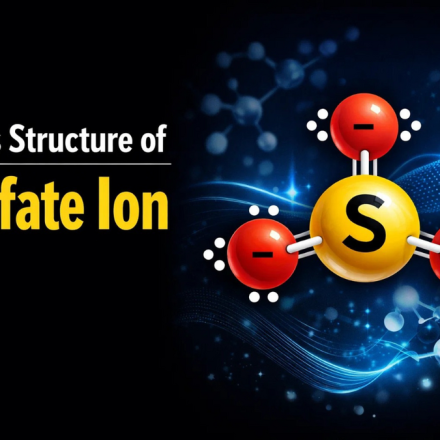
- Lewis structure of sulfate ion has sulfur (S) at the center, bonded to four oxygen (O) atoms, and the overall charge is -2.
- Total valence electrons = 32
- Four S-O bonds are created
- Remaining electrons are distributed as lone pairs on oxygen atoms
- Structure is enclosed in a bracket with a 2- charge
Table Of Content
- Quick Answer (Get It Instantly!)
- Key Highlights
- Lewis Structure of Sulfate Ion Explained Simply
- What is the Lewis Structure of Sulfate Ion?
- Step-by-Step: Sulfate Ion- Lewis Structure Steps
- Step 1: Count the Total Valence Electrons
- Step 2: Determine the Central Atom
- Step 3: Draw Single Bonds
- Step 4: Complete Octet of Oxygen
- Step 5: Check Formal Charges
- Step 6: Understand Resonance
- Step 7: Add Brackets and Charge
- Final Structure Summary
- Why This Structure Matters in Real Life
- Real-World Applications
- Best Practices to Master Lewis Structures
- Common Mistakes Students Make
- Quick Revision Table
- Still Confused? Think Like This
- Conclusion
Key Highlights
- Central atom = Sulfur (S)
- Total valence electrons = 32
- Structure = Tetrahedral geometry
- Includes resonance structures
- Each oxygen has a partial negative charge
- Important for exams, chemistry basics, and molecular understanding
Lewis Structure of Sulfate Ion Explained Simply
Let’s be honest, the Lewis structure is a bit confusing at first. Too many electrons, bonds, and rules.
But, trust us, once you understand the steps, it’s quite simple.
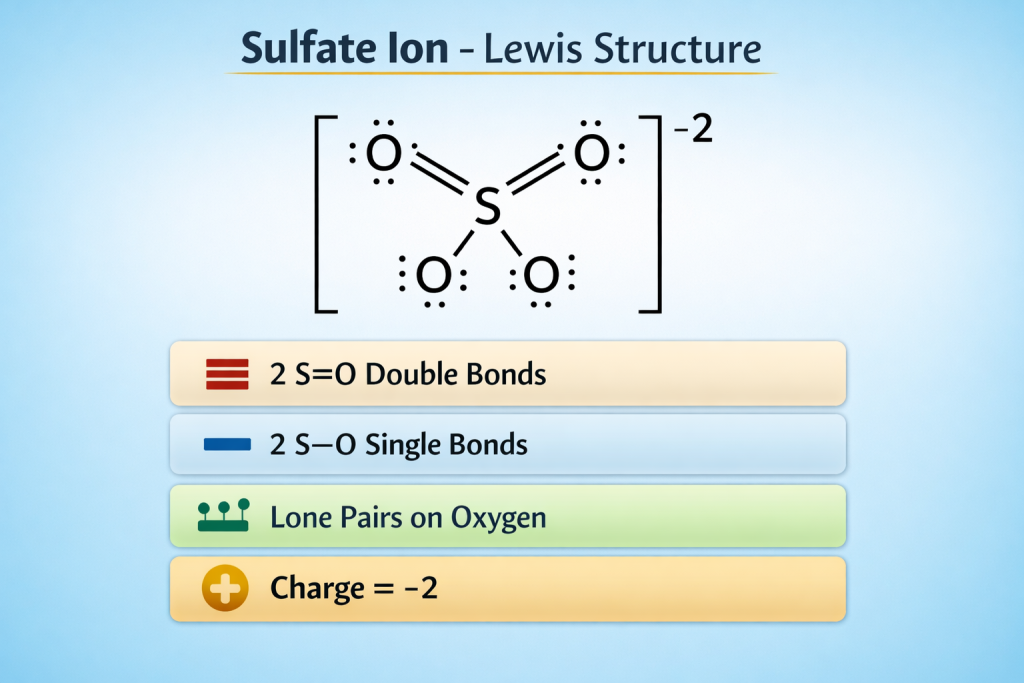
What is the Lewis Structure of Sulfate Ion?
The Lewis structure of sulfate ion is a structure or diagram which shows the arrangement of the electrons between the sulfur and the oxygen atom.
It helps you understand:
- Bond Formation
- Electron Arrangement
- Shape of the molecule
It’s like a map of the electrons.
Step-by-Step: Sulfate Ion- Lewis Structure Steps
Step 1: Count the Total Valence Electrons
- Sulfur (S) – 6
- Oxygen (O) – 6 x 4 = 24
- (charge -2) – +2
Total – 6 + 24 + 2 = 32
Step 2: Determine the Central Atom
Sulfur is less electronegative than oxygen
Sulfur becomes the central atom
Step 3: Draw Single Bonds
Connect the sulfur atom to four oxygen atoms
4 bonds – 8 electrons used
Step 4: Complete Octet of Oxygen
- Oxygen will have lone pairs
- Total electrons used so far = 32
Step 5: Check Formal Charges
This is where many people get confused.
To avoid charges:
- Two oxygen will have a double bond
- Two oxygen will have a single bond
Step 6: Understand Resonance
The two double bonds can be on different oxygen.
This is called resonance
Why it is important:
- It stabilizes the molecule
- The real molecule is a combination of all forms
Step 7: Add Brackets and Charge
Sulfate is an ion.
The structure is put in square brackets with a charge of -2
Final Structure Summary
- 2 S=O Double Bonds
- 2 S–O Single Bonds
- Lone Pairs on Oxygen
- Charge = -2
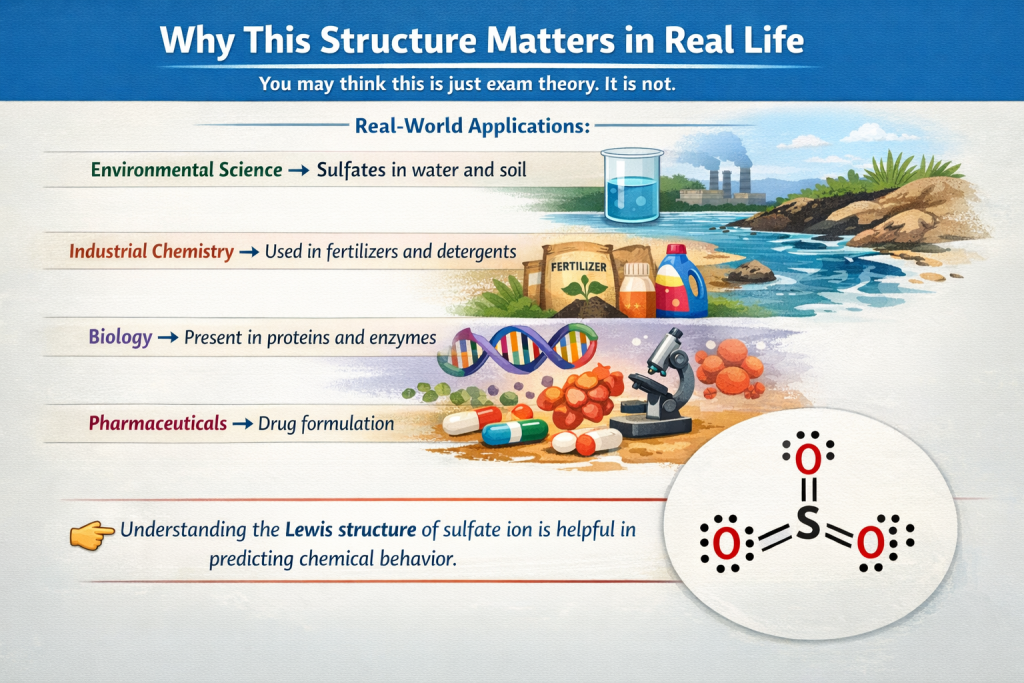
Why This Structure Matters in Real Life
You may think this is just exam theory. It is not.
Real-World Applications:
- Environmental Science → Sulfates in water and soil
- Industrial Chemistry → Used in fertilizers and detergents
- Biology → Present in proteins and enzymes
- Pharmaceuticals → Drug formulation
Understanding the Lewis structure of sulfate ion is helpful in predicting chemical behavior.
Best Practices to Master Lewis Structures
If you’re preparing for exams or interviews, do this:
Count the valence electrons correctly
Determine the right central atom to use
Apply the octet rule correctly
Ensure minimal formal charge is used
Never forget to use resonance
Common Mistakes Students Make
Let’s fix common mistakes early:
- Forgetting to add extra electrons for charge
- Not using the right central atom
- Not using resonance structures correctly
- Not using the octet rule correctly
Making mistakes results in losing marks.
This is a webpage that explains Lewis structures.
Quick Revision Table
| Concept | Value |
|---|---|
| Total electrons | 32 |
| Central atom | Sulfur |
| Shape | Tetrahedral |
| Bonds | 2 double, 2 single |
| Charge | -2 |
Still Confused? Think Like This
Think of Sulfur as a “hub” and “oxygen” as a “connection.”
Some connections are stronger (double bonds), some connections are lighter (single bonds).
And the “electrons”?
Shared resources.
Conclusion
First off, the Lewis structure of the sulfate ion looks pretty complex. But when you follow the steps outlined in the “Sulfate Ion – Lewis Structure” section of this guide, you’ll see how everything makes sense.
You don’t have to memorize this.
You have to see the pattern.
Count the electrons
Draw the bonds
Adjust the charges
Verify the stability