Strongest to Weakest Bonds Order
Strongest to weakest bonds order — here’s the direct answer you’re looking for:
Table Of Content
- 🚀 Key Highlights
- 🧪 Strongest to Weakest Bonds Order Explained Clearly
- ✅ Final Order
- 🔬 Order of Bond Strength in Chemistry (Why This Order Exists)
- 🔷 1. Covalent Bond (Strongest Bond)
- 💡 Example
- 🌍 Real-Life Insight
- 🔷 2. Ionic Bond
- 💡 Example
- 🌍 Real-Life Insight
- 🔷 3. Hydrogen Bond
- 💡 Example
- 🌍 Real-Life Insight
- 🔷 4. Van der Waals Forces (Weakest)
- 💡 Example
- 🌍 Real-Life Insight
- ⚠️ Common Mistakes Students Make
- 💡 Best Practice to Remember Bond Order
- 🌍 Real-World Applications of Bond Strength
- 🧬 Biology
- 🏭 Industry
- 🌡️ Physics & Chemistry
- 🔗 Helpful Resources
- 🌐 External Links
- 🤔 Quick Self-Test
- 🎯 Final Thoughts
👉 Covalent bond > Ionic bond > Hydrogen bond > Van der Waals forces
Yes, that’s the exact strongest to weakest bonds order used in chemistry exams and real-world applications.
If you’ve ever wondered why this order exists or how to remember it without confusion, you’re in the right place. Many students get stuck at this point because they try to memorize instead of understanding the logic behind bond strength.
Let’s break it down in a way that actually makes sense.
🚀 Key Highlights
- Strongest bond = Covalent bond
- Weakest bond = Van der Waals forces
- Bond strength is based on the sharing and attraction of electrons
- Hydrogen bonds exhibit intermediate forces
This knowledge will be helpful during chemistry exams and in practical life
🧪 Strongest to Weakest Bonds Order Explained Clearly
✅ Final Order
- Covalent bond (Strongest)
- Ionic bond
- Hydrogen bond
- Van der Waals forces (Weakest)

🔬 Order of Bond Strength in Chemistry (Why This Order Exists)
To understand the order of bond strength in chemistry, you need to remember one thing:
👉 The greater the attraction between atoms, the greater the bond strength
We will learn about each type of bond.
🔷 1. Covalent Bond (Strongest Bond)
- What happens in this bond?
- Atoms share electrons
Strong attraction between the nuclei and shared electrons
💡 Example
- Water (H₂O)
- Oxygen (O₂)
🌍 Real-Life Insight
Covalent bonds are the backbone of:
- DNA molecules
- Proteins in your body
- Plastic and polymers
👉 That’s why they must be strong – they hold life together.
🔷 2. Ionic Bond
- What happens here?
- One atom loses electrons
- Another atom gains electrons
- Attraction between positive and negative ions
💡 Example
Sodium chloride (NaCl)
🌍 Real-Life Insight
- Ionic compounds:
- Form crystals
- Conduct electricity when dissolved
👉 Used in table salt, batteries, and electrolytes
🔷 3. Hydrogen Bond
- What happens here?
- Attraction between:
- Hydrogen atom
- Electronegative atom (O, N, F)
💡 Example
Water molecules (H₂O)
🌍 Real-Life Insight
Hydrogen bonding explains:
- Why water has high boiling point
- Why DNA strands stay together
👉 Without hydrogen bonds, life wouldn’t exist as we know it.
🔷 4. Van der Waals Forces (Weakest)
✔ What happens here?
Temporary attraction due to movement of electrons
💡 Example
- Noble gases
- Non-polar molecules
🌍 Real-Life Insight
These are weak forces, but they are vital:
- Help gecko stick to walls
- Affect boiling points of gases
⚠️ Common Mistakes Students Make
Let’s face it: these mistakes are extremely common:
❌ Believing ionic bonds have the highest strength
❌ Not taking into account IMFs
❌ Getting hydrogen bonds mixed up with covalent bonds
❌ Not grasping the concept of electrons

💡 Best Practice to Remember Bond Order
Here’s the trick you should remember:
👉 “C I H V”
- C – Covalent
- I – Ionic
- H – Hydrogen
- V – Van der Waals
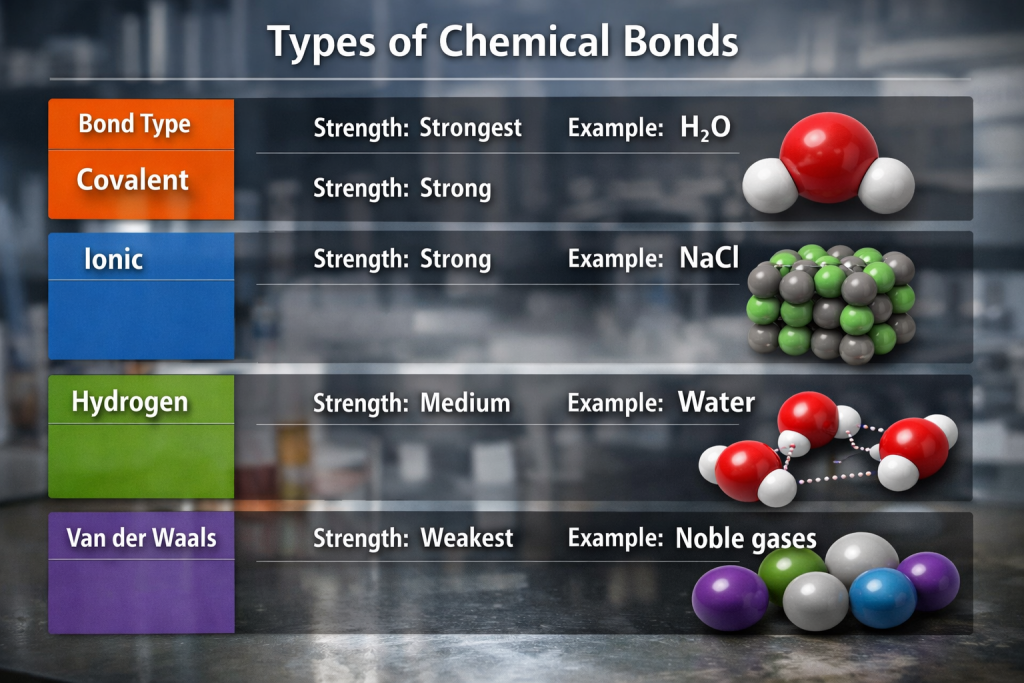
📊 Quick Comparison Table
| Bond Type | Strength | Example |
|---|---|---|
| Covalent | Strongest | H₂O |
| Ionic | Strong | NaCl |
| Hydrogen | Medium | Water |
| Van der Waals | Weakest | Noble gases |
🌍 Real-World Applications of Bond Strength
The order of the strongest, then the weaker, bonds does not remain confined to theory.
🧬 Biology
The structure of DNA relies on Hydrogen bonds
🏭 Industry
Materials rely on Covalent & Ionic bonds
🌡️ Physics & Chemistry
Boiling points rely on Intermolecular forces
📊 Fact: Compounds with strong Covalent bonds have higher Melting points than those with weak Intermolecular forces.
🔗 Helpful Resources
🌐 External Links
🤔 Quick Self-Test
Why are Covalent bonds stronger than Ionic bonds?
Why does Hydrogen bonding play a vital role in the case of Water?
Why are Van der Waals forces weak?
👉 Try it yourself, as learning does.
🎯 Final Thoughts
Strongest, then Weaker, bonds order is something you must not memorize.
Once you grasp the basic concepts of:
🔹 How electrons behave
🔹 What kind of forces are at play
🔹 How molecules interact
👉 Chemistry makes perfect sense, rather than confusing you.
At first, you might find it all too confusing. So many new words. So many new rules.
But when you connect the dots…
👉 It all makes perfect sense.