Valence Electrons of Magnesium
Valence Electrons of Magnesium, Quick Answer First.
Table Of Content
- ✨ Key Highlights
- 📘 What Are Valence Electrons?
- Step 1: Know the Atomic Number
- Final Answer
- 🔹 Group Rule
- 📈 Where This Concept is Used
- 🔹 1. NEET & Board Exams
- 🔹 2. Chemical Bonding
- 🔹 3. Material Science
- 🔹 4. Biology
- ⚙️ Best Practices to Remember
- 🔗 Internal Learning Links
- 🌍 External References
- ❓ Common Questions
- ❓ What are the number of valence electrons in magnesium?
- ❓ What is the reason why magnesium loses electrons?
- ❓ Is magnesium reactive?
- 🧠 Quick Comparison Table
- 🚀 Final Takeaway
- 💬 Conclusion
In case you are looking to find the number of electrons of magnesium, which are considered to belong to the outer electron shell:
👉 Magnesium contains 2 valence electrons.
These are electrons that are located in the outermost shell and they contribute to all chemical reactions of magnesium.
✨ Key Highlights
- ✅ **Mg 2 electrons = 2 valence electrons of magnesium = 2 electrons **
- ✅ Atomic number of magnesium = 12
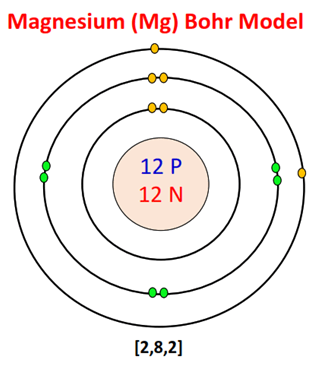
- ✅ Electronic configuration = 2, 8, 2
- ✅ Magnesium is a member of the alkaline earth metals of Group 2.
- ✅ It is likely to lose 2 electrons to become stable.
- ✅ Valency of magnesium = +2
📘 What Are Valence Electrons?
You must first know the meaning of valence electrons in magnesium because it is a concept that cannot be fully understood without first knowing the meaning of the valence electrons of magnesium.
electrons are electrons that are found on the outermost shell of an atom.
These electrons:
- Be involved in chemical bonding.
Choose the degree of reaction of an element. - Establish oxidation and valency.
Compare them to the decision-makers of an atom.
How many electrons of valence does magnesium have?
We will take this one step at a time so you will never forget.
Step 1: Know the Atomic Number
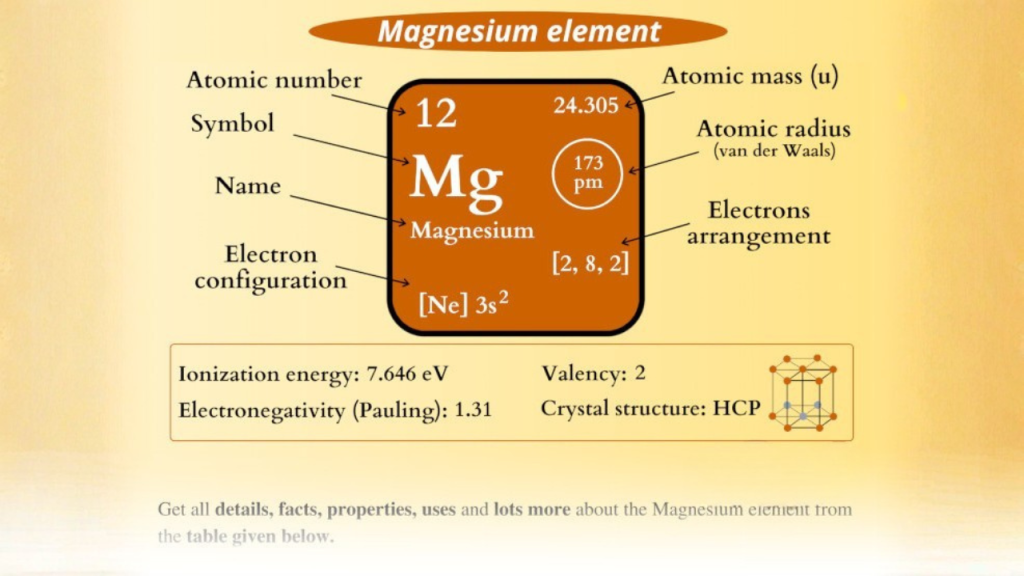
Magnesium is possessing an atomic number of 12.
👉 That means it has 12 electrons
Write Electronic Configuration: This step involves writing the electronic configuration.
You pass electrons out in shells:
- First shell → 2 electrons
- Second shell → 8 electrons
- Third shell → 2 electrons
Hence the set up is:
👉 2, 8, 2
Identifying the Outermost Shell is the 3 rd step.
👉 The shell that is in the outer is the third shell.
👉 It contains 2 electrons
Final Answer:
✔️ 2 = Valence electrons of magnesium.
The reason why Magnesium has 2 valence electrons is explained below.
Magnesium is found in the periodic table under Group 2.
indicating that all elements in Group 2 possess 2 valence electrons.
This is not an accident it is a trend.
🔹 Group Rule:
- Group 1 → 1 valence electron
- Group 2 → 2 valence electrons
- Group 13 → 3 valence electrons
and not to be computed, you can guess:
Group 2 = Magnesium: 2 electrons of valence.
In the case of analogous items, the distinction between the two lies in whether their similarity relies on a particular attribute or is expressed through other means.<|human|>In the real-world analogy, the difference between the two is whether the similarity of the analogous items is based on a specific attribute or is conveyed in a different way.
Imagine this:
There is 2 rupees in your pocket (outer shell electrons).
You are more likely to:
👉 Give away ₹2 completely
rather than attempting to control it partially.
It is precisely what magnesium does:
👉 It loses 2 electrons
👉 and becomes stable as a noble gas (Neon).
What do the following things happen with magnesium that loses 2 electrons?
Magnesium forms:
👉 Mg²⁺ ion
This means:
- It loses 2 electrons
Achieves stable configuration (such as: Neon: 2,8)
This is why magnesium is:
- Highly reactive
Common in ionic compounds (such as MgO, MgCl 2)
📈 Where This Concept is Used
It may seem to you elementary–but it is everywhere:
🔹 1. NEET & Board Exams
Direct questions like:
👉 “What are the number of valence electrons of magnesium?
🔹 2. Chemical Bonding
Helps predict:
- Ionic bonds
- Compound formation
🔹 3. Material Science
Magnesium is used in:
- Lightweight alloys
- Aerospace engineering
🔹 4. Biology
Magnesium plays a role in:
- Chlorophyll (plants)
- Enzyme activation
These concepts are confusing without knowledge of the concept of valence electrons.
⚙️ Best Practices to Remember
To prevent errors in exams:
- ✔️ Atomic number should always be checked first.
- ✔️ Compose electronic configuration in a readable manner.
- ✔️ Identify outermost shell
- ✔️ Use group number shortcut
🔗 Internal Learning Links
You must also learn: to make your fundamentals strong.
Electronic configuration rules.
- Periodic table trends
Oxidation and valency. - Ionic vs covalent bonding
🌍 External References
To learn more, consider:
Toppers rely on these as their resources.
❓ Common Questions
❓ What are the number of valence electrons in magnesium?
👉 2
❓ What is the reason why magnesium loses electrons?
👊 To attain stable noble gas structure.
What is the number of valence of magnesium?
👉 +2
❓ Is magnesium reactive?
Yes it can easily lose 2 electrons.
🧠 Quick Comparison Table
| Element Number of electrons in the atom Atomic number is the count of electrons in the atom’s outer electron shell.< | human | > | Element Number of electrons in the atom Atomic number is the number of electrons in the outer electron shell of the atom. |
|---|---|---|---|
| Magnesium | 12 | 2, 8, 2 | 2 |
| Sodium | 11 | 2, 8, 1 | 1 |
| Aluminum | 13 | 2, 8, 3 | 3 |
🚀 Final Takeaway
Then, in order to forget nothing:
👉ekenelektrons magnesium = 2.
It is a mere concept that enables you to think:
- Chemical bonding
- Reactivity
- Exam questions
💬 Conclusion
It may be overwhelming when you first learn of the atomic structure. Such terms as electronic configuration and valence electrons can be complicated to hear.
But once you understand this:
👉 Surface electrons = valence electrons.
👉 2 electrons outer shell = magnesium.
Everything becomes simple.
One of those core concepts that opens bigger topics in chemistry is the so-called valence electrons of magnesium. This is a solid foundation to build upon and the others become a lot easier.